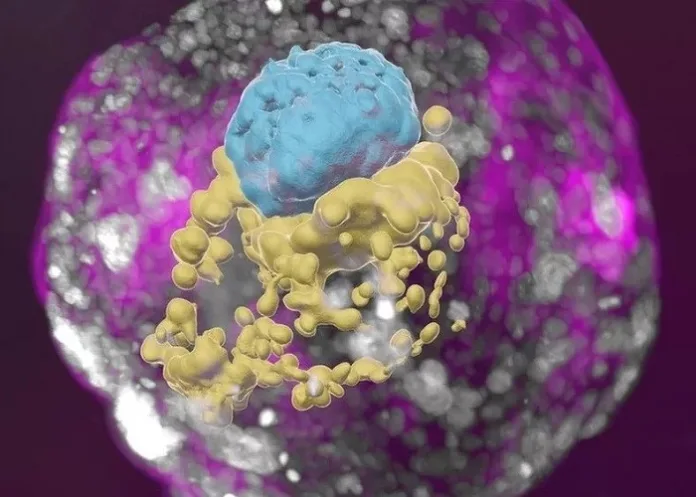
Scientists have grown an entity that closely resembles an early human embryo, without using sperm, eggs or a womb, describing it as the first “complete” embryo model for mimicking all the key structures that emerge in the early embryo.
Israel’s Weizmann Institute of Science team said the model was creating using stem cells, and looks like a textbook example of a real 14-day-old embryo. It even released hormones that turned a pregnancy test positive in the lab, reports BBC News.
The ambition is to provide an ethical way of understanding the earliest moments of our lives, said the team.
The first weeks after a sperm fertilises an egg is a period of dramatic change – from a collection of indistinct cells to something that eventually becomes recognisable on a baby scan.
This crucial time is a major source of miscarriage and birth defects but poorly understood.
“It’s a black box and that’s not a cliché. Our knowledge is very limited,” Professor Jacob Hanna from the team told the BBC.
The research as published in Nature.
Starting material
Embryo research is legally, ethically and technically fraught. But there is now a rapidly developing field mimicking natural embryo development.
“This is really a textbook image of a human day-14 embryo,” Hanna said, “and hasn’t been done before.”
Instead of a sperm and egg, the starting material was naive stem cells, reprogrammed to gain the potential to become any type of tissue in the body.
Chemicals were then used to coax these stem cells into becoming four types of cell found in the earliest stages of the human embryo:
• epiblast cells, which become the embryo proper (or foetus);
• trophoblast cells, which become the placenta;
• hypoblast cells, which become the supportive yolk sac; and
• extraembryonic mesoderm cells.
A total of 120 of these cells were mixed in a precise ratio – and then, the scientists stepped back and watched.
About 1% of the mixture began the journey of spontaneously assembling themselves into a structure that resembles, but is not identical to, a human embryo.
“I give great credit to the cells – you have to bring the right mix and have the right environment and it just takes off,” Hanna said. “That’s an amazing phenomenon.”
The embryo models were allowed to grow and develop until they were comparable to an embryo 14 days after fertilisation. In many countries, this is the legal cut-off for normal embryo research.
‘Making sense’
The hope is embryo models can help scientists explain how different types of cell emerge, witness the earliest steps in building the body’s organs or understand inherited or genetic diseases.
Already, this study shows other parts of the embryo will not form unless the early placenta cells can surround it.
There is even talk of improving in vitro fertilisation (IVF) success rates by helping to understand why some embryos fail or using the models to test whether medicines are safe during pregnancy.
Professor Robin Lovell Badge, who researches embryo development at the Francis Crick Institute, London, said these embryo models “look pretty good and pretty normal”.
“I think it’s done very well… it’s all making sense and I’m impressed,” he said.
But the current 99% failure rate would need to be improved, he added. It would be hard to understand what was going wrong in miscarriage or infertility if the model failed to assemble itself most of the time.
Legally distinct
The work also raises the question of whether embryo development could be mimicked past the 14-day stage.
This would not be illegal, as embryo models are legally distinct from embryos.
“Some will welcome this, but others won’t like it,” Lovell-Badge said.
And the closer these models come to an actual embryo, the more ethical questions they raise.
They are not normal human embryos, they’re embryo models, but they’re very close to them.
“So should you regulate them in the same way as a normal human embryo or can you be a bit more relaxed about how they’re treated?”
Professor Alfonso Martinez Arias, from the department of experimental and health sciences at Pompeu Fabra University in Barcelona, Spain, said it was “a most important piece of research”.
“The work has… achieved a faithful construction of the complete structure (of a human embryo) from stem cells in the lab, thus opening the door for studies of the events that lead to the formation of the human body plan,” he said.
The researchers stress it would be unethical, illegal and actually impossible to achieve a pregnancy using these embryo models. Assembling the 120 cells together goes beyond the point an embryo could successfully implant into the lining of the womb.
Study details
Complete human day 14 post-implantation embryo models from naïve ES cells
Bernardo Oldak, Emilie Wildschutz, Vladyslav Bondarenko, Jacob Hanna, et al.
Published in Nature on 6 September 2023
Abstract
The ability to study human post-implantation development remains limited due to ethical and technical challenges associated with intrauterine development after implantation. Embryo-like models with spatially organised morphogenesis of all defining embryonic and extra-embryonic tissues of the post-implantation human conceptus (i.e., embryonic disk, bilaminar disk, yolk- and chorionic sacs, surrounding trophoblasts) remain lacking. Mouse naïve embryonic stem cells (ESCs) have recently been shown to give rise to embryonic and extra-embryonic stem cells capable of self-assembling into post-gastrulation mouse Structured Stem cell-based Embryo Models with spatially organized morphogenesis (SEMs).
Here, we extend these findings to humans, while using only genetically unmodified human naïve ESCs (in HENSM conditions). Such human fully integrated SEMs recapitulate the organisation of nearly all known lineages and compartments of post-implantation human embryos including epiblast, hypoblast, extra-embryonic mesoderm, and trophoblast surrounding the latter layers. These human complete SEMs demonstrated developmental growth dynamics that resemble key hallmarks of post-implantation stage embryogenesis up to 13-14 days’ post-fertilisation (dpf) (Carnegie stage 6a). This includes embryonic disk and bilaminar disk formation, epiblast lumenogenesis, polarised amniogenesis, anterior-posterior symmetry breaking, PGC specification, polarised yolk sac with visceral and parietal endoderm, extra-embryonic mesoderm expansion that defines a chorionic cavity and a connecting stalk, a trophoblast surrounding compartment demonstrating syncytium and lacunae formation. This SEM platform may enable the experimental interrogation of previously inaccessible windows of human early post-implantation up to peri-gastrulation development.
BBC article – Scientists grow whole model of human embryo, without sperm or egg (Open access)
See more from MedicalBrief archives:
Biotech company plans to create embryos for organ harvesting
World's first ‘vagina chip’ designed to better understand women's health